La Bibliothèque d'Applications présente des modèles construits avec COMSOL Multiphysics pour la simulation d'une grande variété d'applications, dans les domaines de l'électromagnétisme, de la mécanique des solides, de la mécanique des fluides et de la chimie. Vous pouvez télécharger ces modèles résolus avec leur documentation détaillée, comprenant les instructions de construction pas-à-pas, et vous en servir comme point de départ de votre travail de simulation. Utilisez l'outil de recherche rapide pour trouver les modèles et applications correspondant à votre domaine d'intérêt. Notez que de nombreux exemples présentés ici sont également accessibles via la Bibliothèques d'Applications intégrée au logiciel COMSOL Multiphysics® et disponible à partir du menu Fichier.
This tutorial explores the current distribution in a low-temperature PEMFC when using serpentine-based flow field patterns. The cell is operated in counter-flow mode so that the oxygen and hydrogen inlet flow streams are located at opposite sides. Relatively dry inlet gas compositions ... En savoir plus
This tutorial models the thermal management of a polymer electrolyte membrane (PEM) fuel cell stack. Operating the stack with a similar temperature profile for all cells is important since an uneven temperature distribution may otherwise result in nonuniform water vapor condensation and ... En savoir plus
This tutorial models the intercoupled electrochemical reactions, charge and species transport as well as heat transfer in a polymer electrolyte membrane (PEM) fuel cell. For the gas flow fields, straight channels are used on the hydrogen anode side, whereas a mesh structure is used on ... En savoir plus
In a polymer electrolyte membrane electrolyzer cell (PEMEC), the two electrode compartments are separated by a polymer membrane. Liquid water is fed to the anode side, forming oxygen gas on the anode, and hydrogen gas on the cathode side, respectively. The respective designs of the ... En savoir plus
This example models a solid oxide electrolyzer cell wherein water vapor is reduced to form hydrogen gas on the cathode, and oxygen gas is evolved on the anode. The current distribution in the cell is coupled to the cathode mass transfer of hydrogen and water and momentum transport. Two ... En savoir plus
The electrochemical cell shown in this model can be regarded as a unit cell of a larger wire-mesh electrode that is common in many industrial processes. One of the most important aspects in the design of electrochemical cells is the current density distributions in the electrolyte and ... En savoir plus
This example models the flow and mass transport in the channels and the gas diffusion layer (GDL) of a polymer electrolyte fuel cell. The cathode electrode reaction is modeled as a boundary condition, where the local current density depends on the overpotential and the local oxygen ... En savoir plus
In an alkaline electrolyzer stack, all cells share the same electrolyte. As a result of all cells being in ionic contact, parasitic shunt currents flow between the cells through the manifolds and the electrolyte channels, on both the inlet and outlet side. This example models a ... En savoir plus
This model defines a zero-gap alkaline water electrolyzer, where oxygen and hydrogen gas are evolved in porous gas diffusion nickel felt electrodes, placed adjacent to a porous separator (diaphragm). The geometry defines a unit cell of an electrolyzer stack, in turn comprising two full ... En savoir plus
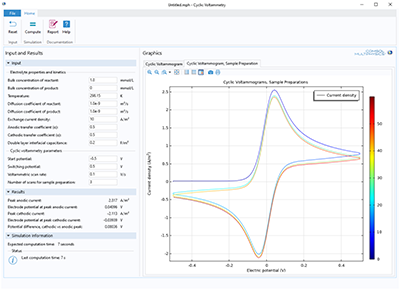
The purpose of the app is to demonstrate and simulate the use of cyclic voltammetry. You can vary the bulk concentration of both species, transport properties, kinetic parameters, as well as the cycling voltage window and scan rate. Cyclic voltammetry is a common analytical technique ... En savoir plus