
Récemment, Tesla Inc, un important fabricant de voitures électriques et de batteries, a annoncé un nouveau concept de design “sans languettes” (tabless) pour les batteries lithium-ion cylindriques. Cela a suscité un certain émoi parmi les experts de la batterie. (Ref. 1) Tesla affirme que cette innovation permet d’augmenter considérablement l’autonomie et la puissance de cellules basées sur des chimies déjà existantes. Pour une communauté habituée à des progrès annuels se limitant à un pourcentage à un chiffre, cela semblait presque trop beau pour être vrai. Cependant, en creusant la physique sous-jacente des batteries, on découvre rapidement pourquoi ce concept pourrait permettre d’augmenter le kilométrage de nos futurs véhicules électriques. Dans cet article de blog, nous nous penchons sur cette nouvelle architecture de connecteurs.
Simuler les batteries en incluant les connecteurs
Reprenons depuis le début : une cellule de batterie au lithium est constituée d’un empilement de différentes couches — feuilles métalliques collectrices de courant, électrodes poreuses et séparateurs — placé dans une enceinte contenant un électrolyte. La configuration de l’empilement dépend du type de boîtier (pile bouton, cellule prismatique, cylindrique ou poche, etc.). Les batteries lithium-ion cylindriques sont fabriquées en enroulant les différentes couches de la batterie sous forme d’un cylindre, qui est ensuite placé dans un boîtier métallique. La structure en spirale qui en résulte est communément appelée “jelly roll”, en raison de sa ressemblance avec la pâtisserie d’Europe centrale.
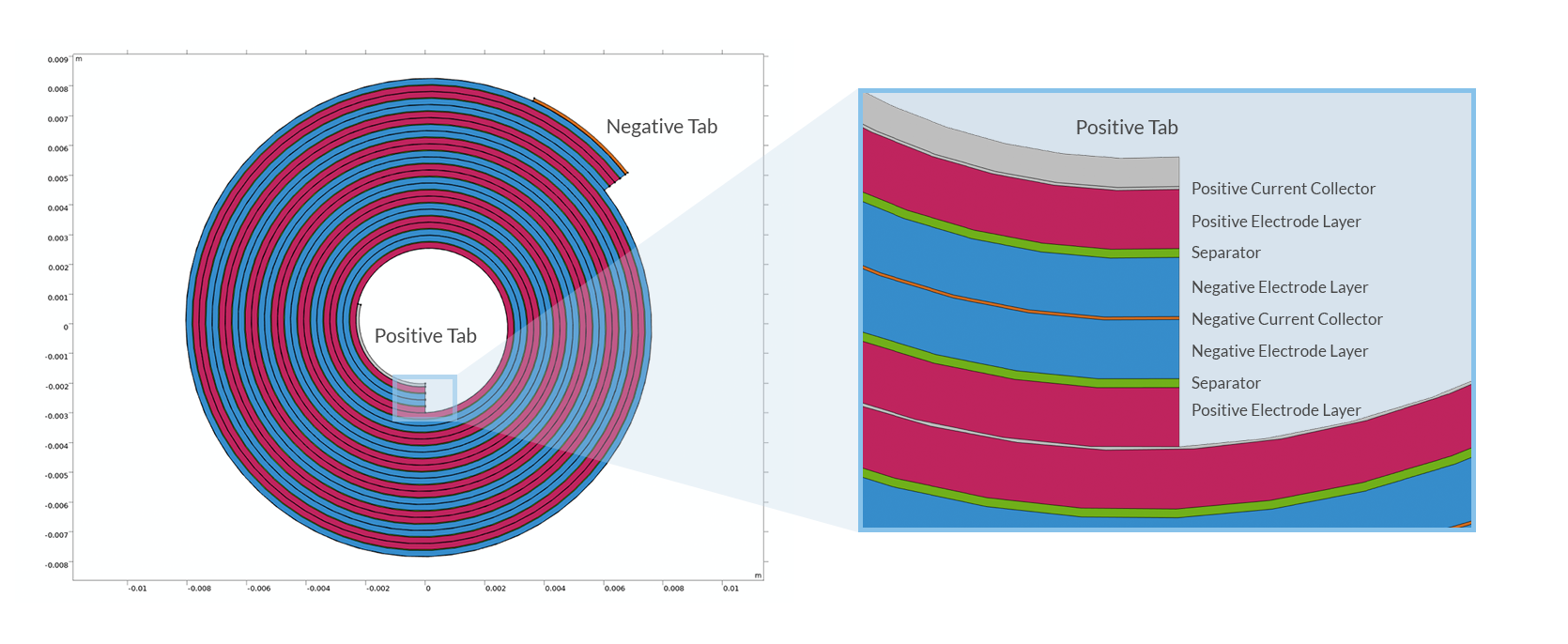
Coupe transversale d’un enroulement en spirale, ou jelly roll, dans une batterie cylindrique.
Les collecteurs de courant sont généralement des feuilles métalliques constitués de cuivre (électrode négative) et d’aluminium (électrode positive), toutes deux d’une épaisseur d’environ quelques dizaines de micromètres, voire moins. Le choix des métaux s’explique par leur stabilité électrochimique à différents potentiels. Afin de faire circuler le courant du jelly roll vers l’extérieur, jusqu’au boitier, des bandes de métal supplémentaires, ou languettes, sont soudées aux collecteurs de courant. Le design le plus simple de ces connecteurs, utilisé dans les piles de faible puissance, place une languette de chaque côté de l’enroulement.
Une première approche pour étudier l’effet des connecteurs consiste à créer un modèle pseudostationnaire qui tient compte des pertes de tension ohmiques dans les conducteurs électroniques et dans l’électrolyte, ainsi que des surtensions d’activation dues aux réactions de transfert de charge dans les électrodes. La distribution de courant interne de la batterie qui résulte de ce modèle est appelée distribution de courant secondaire par les électrochimistes. Comme ce modèle ne tient pas compte de l’accumulation ou de la perte d’atomes de lithium dans les électrodes ou des ions lithium dans l’électrolyte, il peut être considéré comme une vue instantanée du fonctionnement d’une batterie à un certain état de charge. En réalité, la concentration locale change au fil du temps, au fur et à mesure que la batterie est chargée ou déchargée. Toutefois, un modèle de distribution de courant secondaire peut prédire avec précision les pertes de tension dans le jelly roll à un moment donné et à un état de charge donné.
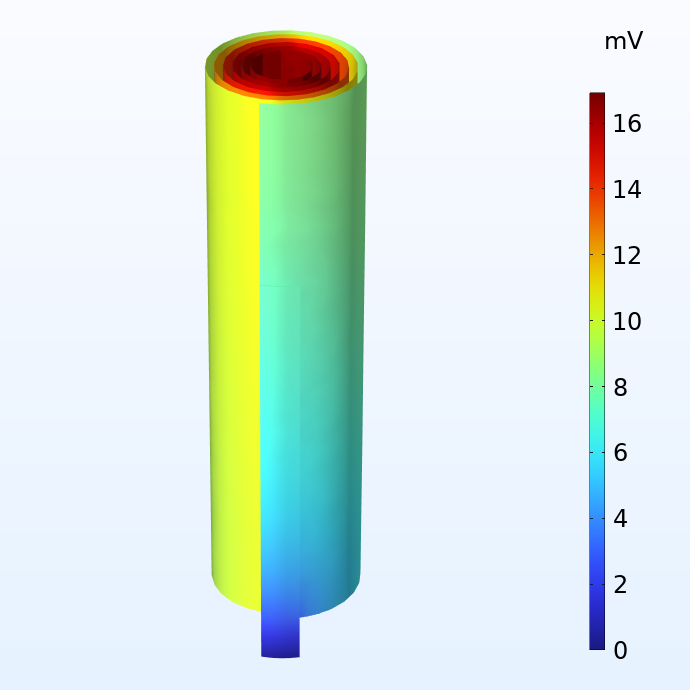
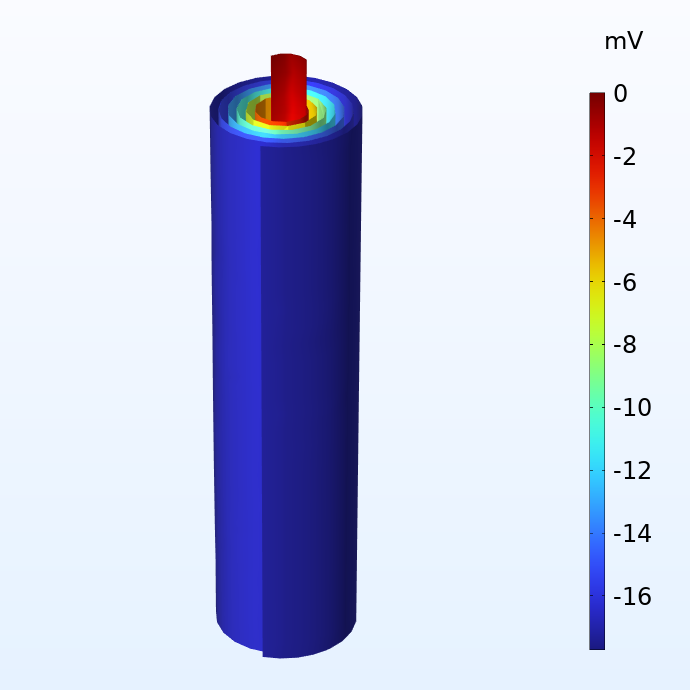
Potentiel dans les collecteurs de courant négatif (à gauche) et positif (à droite) par rapport aux terminaux de courant correspondants.
La figure ci-dessus montre la distribution du potentiel dans les collecteurs de courant négatif et positif d’un jelly roll lorsque celui-ci est soumis à une décharge 1C, ce qui correspond au courant équivalent nécessaire pour charger ou décharger la batterie en une heure. La taille du jelly roll correspond au boîtier d’une batterie 18650, qui mesure 65 mm de haut et 18 mm de diamètre. Nous constatons que, bien que les pertes de potentiel dans les collecteurs de courant soient relativement faibles, elles ne sont pas négligeables. Pour une cellule plus grande comme la cellule 4680 (80 mm de haut et 46 mm de diamètre) que Tesla prévoit de fabriquer, les pertes de tension deviendraient significatives si l’on utilisait un design traditionnel des connecteurs.
Notre modèle étant basé sur la physique, les sources de chaleur locales peuvent être facilement dérivées sur la base des pertes ohmiques (chauffage par effet Joule) et des surtensions d’activation. En introduisant notre variable de source de chaleur dans un modèle de transfert de chaleur, nous obtenons les résultats suivants.
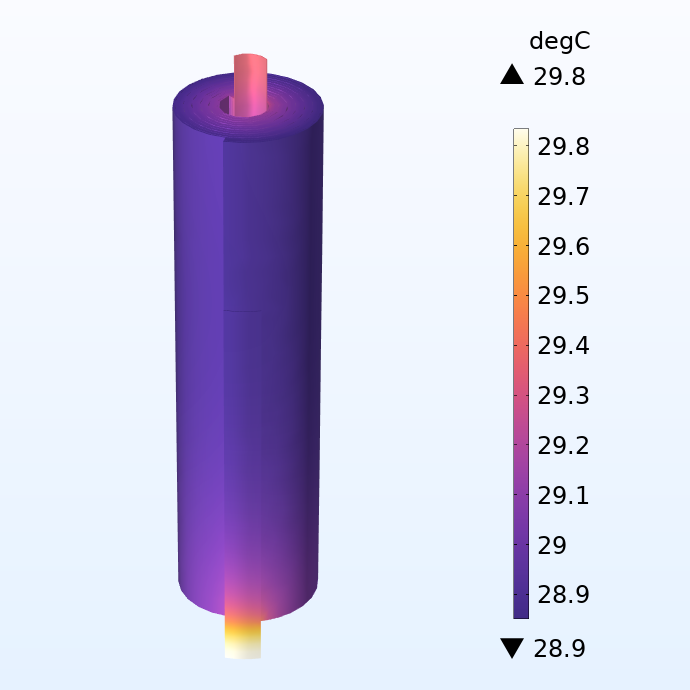
Distribution de la température dans le jelly roll.
Nous appliquons ici une condition de refroidissement par convection sur la zone extérieure de l’enroulement, en imposant un flux de chaleur proportionnel à la différence entre la température de surface et la température extérieure (25°C). La chaleur transportée par les bornes électriques aux extrémités des languettes est négligée.
Si l’on examine la distribution de la température, on constate une forte augmentation de la température dans les languettes. Cela indique que le chauffage par effet Joule dans les languettes entraîne un échauffement local important dans cette batterie de taille relativement réduite, même pour des courants modérés. Les écarts de température locaux peuvent également se propager dans les couches d’électrodes adjacentes, ce qui entraîne un vieillissement plus rapide de certains éléments de la batterie, limitant ainsi la durée de vie de l’ensemble.
La véritable géométrie du jelly roll se prête mal à la modélisation et la simulation. Par exemple, il peut être délicat de dessiner des objets dans une géométrie en spirale, notamment pour l’ajout de plusieurs languettes à l’intérieur de l’enroulement. En outre, il est difficile de visualiser les résultats au sein des différentes couches, lorsque l’on souhaite par exemple tracer la densité de courant à travers les séparateurs à différents endroits dans l’enroulement.
Modélisation d’un jelly roll aplati
Nous pouvons également définir le même modèle sur une version aplatie (déroulée) de la géométrie du jelly roll. Cela nous permet d’introduire facilement des languettes et de mieux examiner le modèle et les résultats de la simulation. Dans ce cas, nous pouvons enrouler virtuellement la batterie. L’illustration ci-dessous montre une version aplatie du jelly roll où les différentes couches et languettes ont été dessinées sous forme de blocs rectangulaires.

Représentation aplatie de la géométrie en spirale du jelly roll.
En réalité, lors de la création du jelly roll à partir de l’empilement de couches, la face avant (verte) se retrouve en contact avec la face arrière de la pile de blocs rectangulaires (voir la figure ci-dessus). Des conditions limites de couplage spécifiques, appelées couplages non locaux dans le logiciel COMSOL Multiphysics® sont utilisées dans le modèle de jelly roll aplati afin de relier mathématiquement ces frontières désormais détachées géométriquement. C’est ce que nous entendons par “enrouler virtuellement la batterie”.
La géométrie aplatie présente également l’avantage de nécessiter moins d’éléments de maillage, puisqu’il n’est pas nécessaire de résoudre la courbure locale de l’enroulement. En outre, on constate que le profil de température pour la géométrie aplatie reproduit fidèlement les résultats obtenus avec le jelly roll (voir ci-dessus), ce qui indique que cette transformation d’aplatissement n’a que peu d’effets sur les résultats.

Température (°C) dans le jelly roll aplati.
La géométrie aplatie permet maintenant de visualiser facilement la densité de courant à travers les séparateurs, comme le montre la figure ci-dessous.
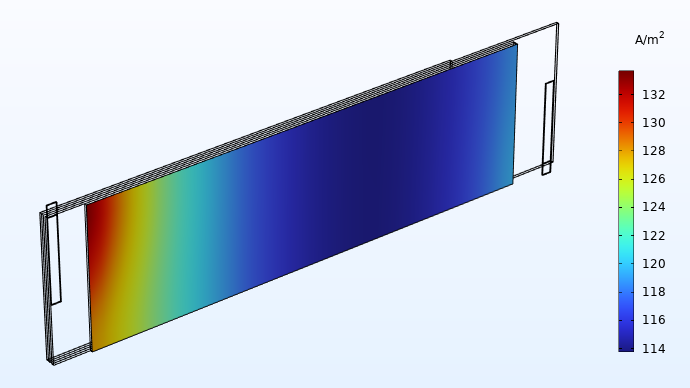
Distribution du courant (A/m2) dans la direction perpendiculaire au plan de l’un des séparateurs.
Les graphiques de distribution de courant comme celui-ci fournissent des informations précieuses aux concepteurs de batterie. Ici, le graphique montre une densité de courant nettement plus élevée dans la zone proche des languettes. Cela signifie que la batterie subit une usure électrochimique plus importante dans les zones les plus proches des languettes, ce qui entraîne un vieillissement accéléré. Si l’on laisse la cellule fonctionner plus longtemps, la distribution de courant illustrées ci-dessus finira par s’équilibrer pour donner un profil plus uniforme. Toutefois, dans le cas d’une batterie soumise à un cycle plus court autour d’un état de charge fixe (par exemple, dans un système de batteries utilisé pour l’écrêtement des pointes de charge), notre évaluation de la distribution du courant ci-dessus est tout à fait pertinente.
Etudier les designs à connecteurs intégrés avec COMSOL Multiphysics®
Utilisons maintenant l’approche de modélisation susmentionnée pour étudier l’architecture dite “sans languettes”.
Ce design consiste à supprimer les languettes métalliques supplémentaires et à laisser les feuilles métalliques des collecteurs de courant conduire le courant vers l’extérieur. Pour ce faire, les feuilles sont prolongées jusqu’à dépasser de la surface des électrodes. Comme ces feuilles sont très fines, il est nécessaire de disposer d’une multitude de ces bandes étendues pour limiter les résistances ohmiques. Le terme “sans languettes” est en fait un peu trompeur, car les languettes ne sont pas entièrement supprimées mais sont intégrées aux collecteurs de courant. C’est pourquoi nous utilisons les termes languettes intégrées et connecteurs intégrés dans la suite de ce billet de blog.
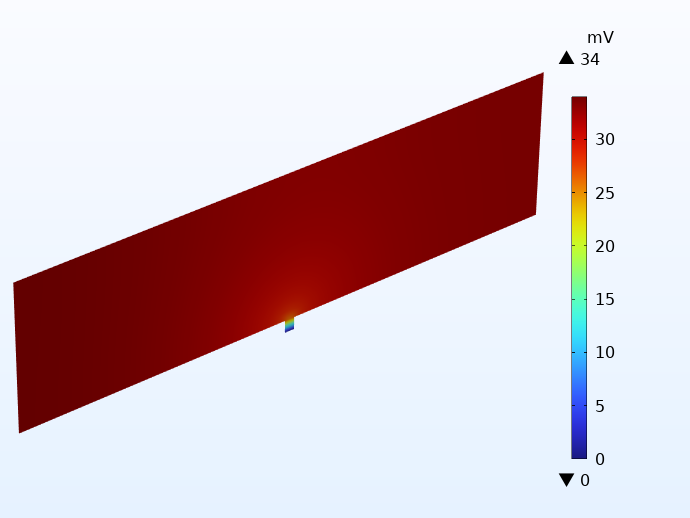
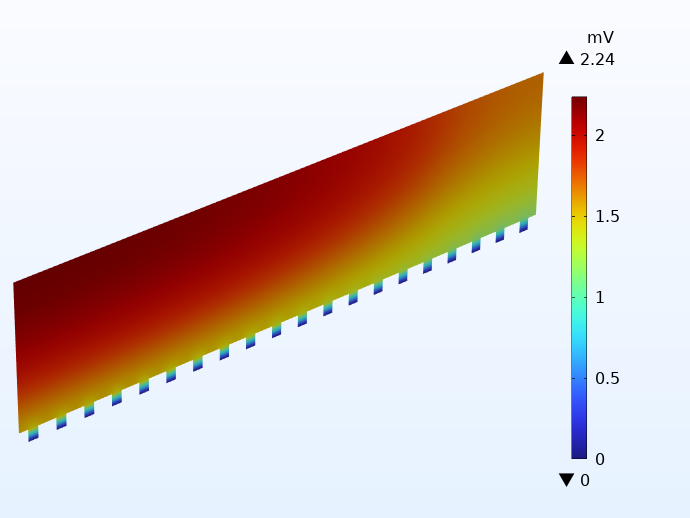
Pour illustrer comment cela fonctionne en pratique, on a représenté ci-dessous la distribution de potentiel sur le collecteur de courant négatif pour deux cellules utilisant des languettes intégrées. En dehors de la configuration des languettes, les cellules sont identiques aux exemples présentés ci-dessus. L’illustration de gauche ne comporte qu’une seule languette intégrée, tandis que l’exemple de droite en compte vingt de chaque côté. Notez les échelles de potentiel. La différence est de plus de 30 mV !
Comparaison du potentiel électrique dans les collecteurs de courant négatifs de deux cellules à connecteurs intégrés : une cellule à 1 languette (à gauche) et une autre cellule à 20 languettes (à droite).
L’utilisation d’une seule languette intégrée produit des pertes de potentiel très élevées, ce qui se traduit par des pertes thermiques locales importantes. En ce qui concerne la cellule intégrant plusieurs languettes, les pertes de potentiel sont inférieures à celles de la cellule à languettes traditionnelles.
L’exemple comprenant une seule languette intégrée doit être considéré comme un cas extrême. Cependant, si nous voulons économiser les matières premières et réduire le poids de la batterie, nous ne devons pas utiliser plus de languettes que nécessaire. Afin d’étudier l’impact du nombre de languettes, nous pouvons effectuer une étude de balayage paramétrique et tracer la surtension totale et la température maximale de la cellule en fonction du nombre de languettes.
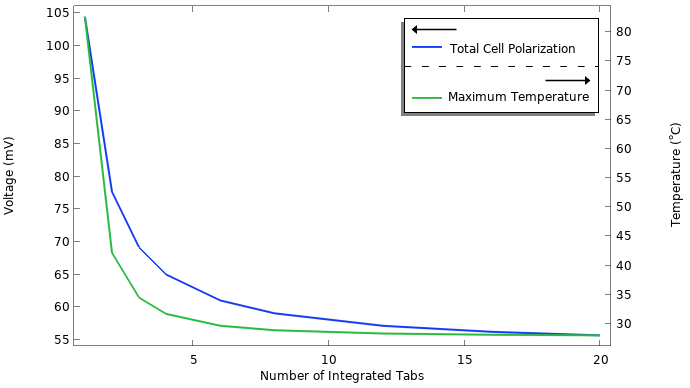
Surtension totale de la cellule et température maximale en fonction du nombre de languettes intégrées.
Comme le montre le graphique, nous nous approchons assez rapidement d’une asymptote. Nous pouvons constater que l’ajout de plus de 20 languettes intégrées n’abaisse pas la surtension de la cellule en dessous du niveau de 55 mV. Cette surtension résiduelle de la cellule résulte d’autres processus qui ne sont pas affectés par le nombre de languettes, tels que la résistance de l’électrolyte et les réactions de transfert de charge. Nous constatons également que l’utilisation d’une ou deux languettes seulement n’est pas envisageable, car les températures atteintes sont très élevées. Or, une température élevée accélère le vieillissement et constitue également un risque direct de sécurité.
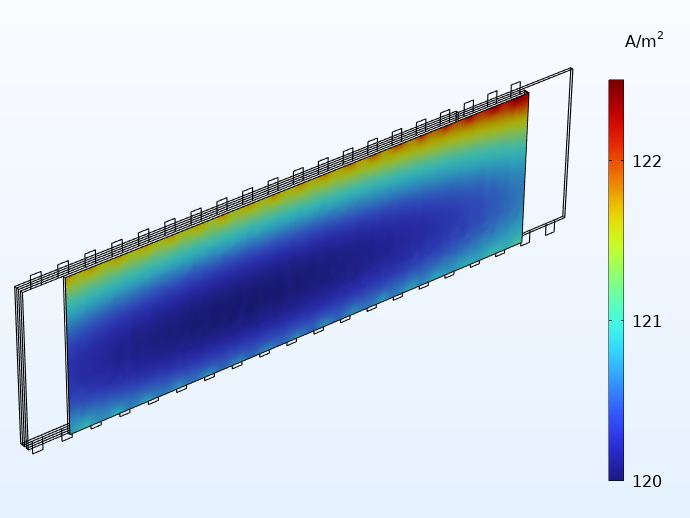
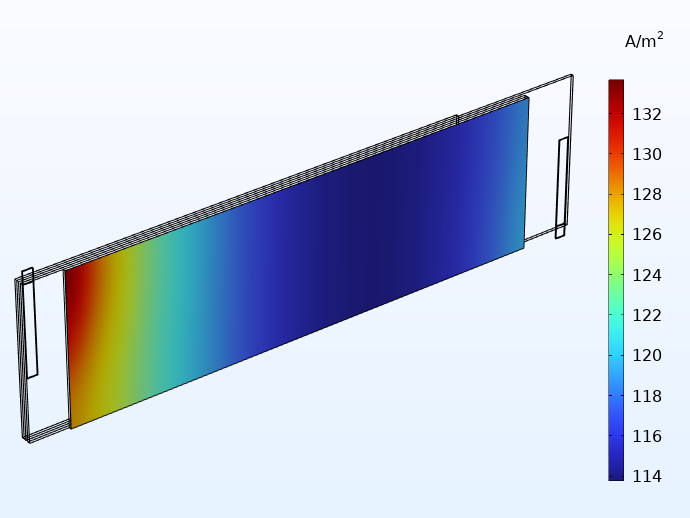
En comparant ci-dessous les distributions de courant obtenues avec les connecteurs intégrés et les connecteurs traditionnels, nous constatons que les languettes intégrées permettent d’obtenir une distribution plus uniforme. En outre, les variations qui subsistent sont désormais principalement observés dans le sens de la hauteur du jelly roll.
Distribution du courant dans la direction perpendiculaire au plan de l’un des séparateurs en considérant 20 languettes intégrées (à gauche) et des languettes traditionnelles (à droite).
En ce qui concerne le transfert de chaleur, les languettes intégrées permettent d’améliorer le refroidissement interne du jelly roll. En effet, les languettes sont en contact thermique avec la surface de refroidissement extérieure par l’intermédiaire d’un excellent conducteur de chaleur, le métal. Chaque languette est en contact thermique et électrique avec l’extérieur.
La figure ci-dessous montre la distribution de température dans le jelly roll à connecteurs intégrés. Nous pouvons voir sur ce graphique que le champ de température est beaucoup plus uniforme qu’avec les connecteurs traditionnels.
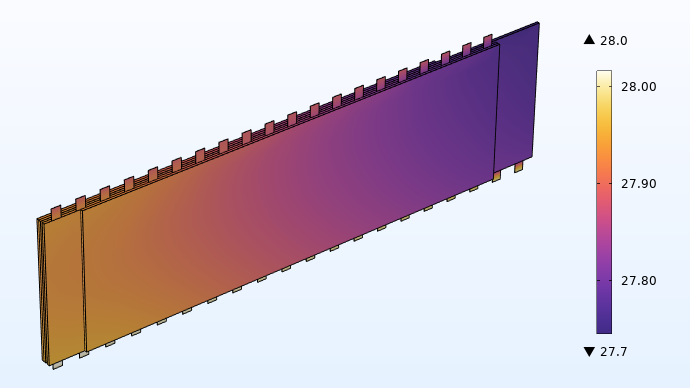
Distribution de la température (°C) avec 20 languettes intégrées.
Pour conclure
En résumé, les languettes intégrées améliorent considérablement la conduction du courant et la conduction de la chaleur vers l’extérieur de la cellule. Il devient ainsi possible de construire des cellules cylindriques de plus grand rayon, ce qui pourrait permettre de produire des packs de batteries ayant une densité d’énergie et de puissance plus élevée. Un rayon de batterie plus grand permet en effet d’obtenir un rapport plus important entre la quantité de matériau interne et l’enveloppe externe (boîtier) de la batterie. Cela explique également les ambitions et les déclarations de Tesla.
Le design dit “sans languettes” s’avère être une alternative innovante aux connecteurs traditionnels des batteries lithium-ion. Toutefois, celui-ci pose également de nouveaux défis techniques qui peuvent être appréhendés à l’aide du logiciel COMSOL Multiphysics®.
Prochaines étapes
- Découvrez comment la méthodologie présentée peut être utile pour vos simulations en téléchargeant ces tutoriels :
- Consultez ces billets de blog connexes :
- View Présentation de Tesla sur le design sans languettes
Référence
- A. J. Hawkins, “Tesla announces ‘tabless’ battery cells that will improve range of its electric cars”, The Verge, 22 Sep. 2020; https://www.theverge.com/2020/9/22/21449238/tesla-electric-car-battery-tabless-cells-day-elon-musk





Commentaires (0)